GASIFICATION AND BIOCHAR FORMATION
Gasification is a thermochemical conversion process characterized by the partial oxidation of biomass under sub-stoichiometric conditions, where a controlled and limited supply of oxidizing agents (λ < 1) is introduced. Unlike pyrolysis, which operates in the complete absence of oxygen, gasification intentionally allows a small amount of oxidant—such as air, pure oxygen, steam, carbon dioxide, or their combinations—to drive a series of exothermic and endothermic reactions. The primary objective of gasification has traditionally been the production of synthesis gas (syngas), a combustible mixture mainly composed of carbon monoxide (CO), hydrogen (H₂), methane (CH₄), and light hydrocarbons. Biochar, in this context, is typically considered a secondary solid by-product, although its production can be strategically enhanced under specific operating conditions.
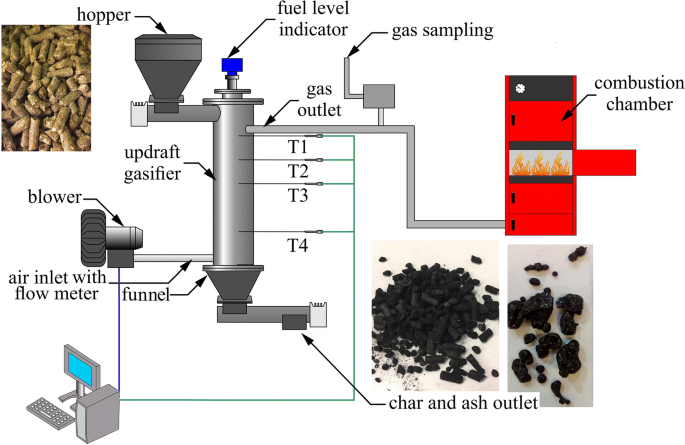
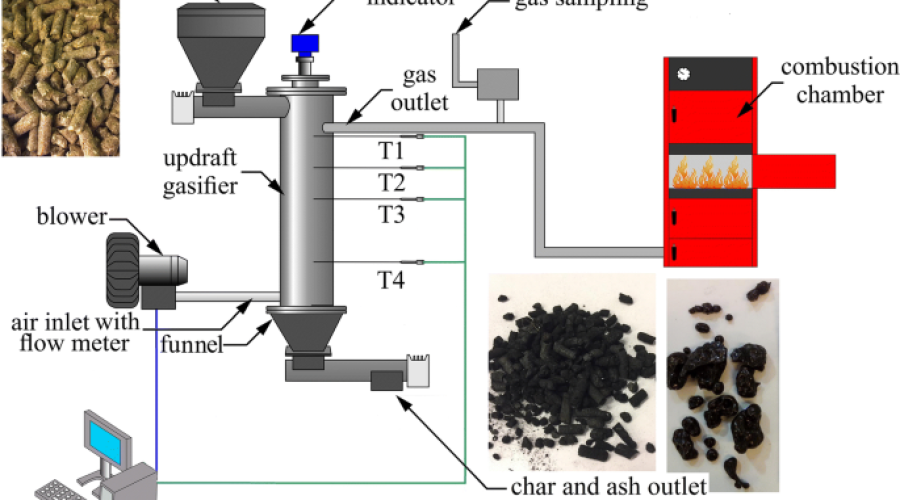
Gasification generally occurs at higher temperatures than pyrolysis, typically within the range of 700 to 1000 °C, where both thermal decomposition and oxidation reactions coexist. The process proceeds through several interconnected stages. Initially, the biomass undergoes drying and devolatilization, similar to pyrolysis. This is followed by partial oxidation, where a fraction of the carbon is combusted to generate heat, sustaining the process energetically. Subsequently, a complex network of heterogeneous gas–solid reactions takes place. These include char oxidation (C + O₂ → CO₂ or CO), steam gasification (C + H₂O → CO + H₂), and the Boudouard reaction (C + CO₂ → 2CO), all of which contribute to the formation and composition of syngas.

Due to the presence of oxidizing agents and the elevated temperatures, gasification typically results in lower solid yields compared to pyrolysis, as a significant portion of the carbon is converted into gaseous products. However, the process can be engineered toward a char-oriented mode when biochar is a target product. This can be achieved by carefully controlling key parameters such as:
- Limiting the oxygen or oxidant supply to suppress complete oxidation
- Reducing the residence time of the solid phase to prevent excessive carbon conversion
- Extracting char from intermediate zones within the reactor before full gasification occurs
Through such optimization, it is possible to balance syngas production with meaningful biochar recovery.
The biochar derived from gasification typically exhibits distinct physicochemical properties compared to pyrolysis-based biochar. Due to the higher thermal severity, it tends to have a higher degree of carbonization, resulting in a more condensed aromatic structure and greater chemical stability. At the same time, it generally contains lower volatile matter, reflecting the more complete removal of labile organic compounds. However, gasification biochar often shows an elevated ash content, as mineral components become concentrated during the process and may also undergo transformation at high temperatures.
These characteristics make gasification-derived biochar particularly suitable for applications requiring high stability and long-term carbon sequestration, although its higher ash content and altered surface chemistry may influence its performance in soil amendment or adsorption applications. From a systems perspective, gasification offers an integrated pathway where energy production (via syngas) and carbon sequestration (via biochar) can be combined, making it highly relevant for circular economy models and carbon-negative technologies.
In summary, while gasification has traditionally been optimized for energy generation, it can be strategically adapted to serve as a dual-purpose process—producing both renewable energy and stable biochar—thereby contributing to sustainable agriculture, waste valorization, and climate mitigation objectives.